 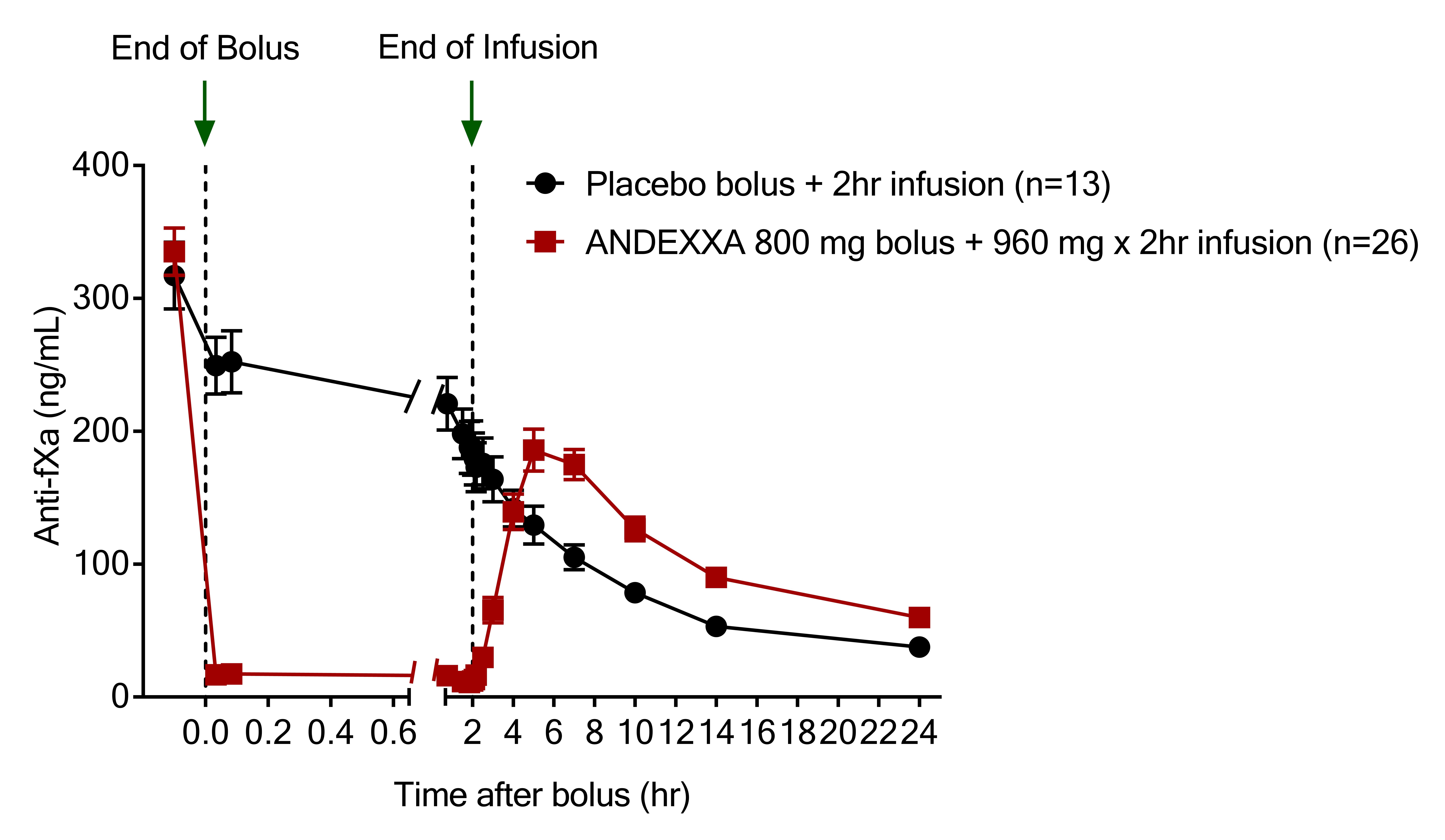
“Today’s approval represents a significant step forward in patient care and one that the medical community has been eagerly anticipating,” Stuart J. Previously, the Portola product had received both Orphan Drug and Breakthrough Therapy designations from the FDA, and it was granted approval under the agency’s Accelerated Approval pathway founded on the change in anti-Factor Xa activity in healthy volunteers from baseline measurements. The US Food and Drug Administration (FDA) has approved andexanet alfa (AndexXa, Portola Pharmaceuticals) as the first and only antidote for patients treated with rivaroxaban and apixaban when the reversal of anticoagulation is needed due to life-threatening or uncontrolled bleeding. 
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
